 Groups (columns) in the f-block (between groups 2 and 3) are not numbered. However, they remain d-block elements even when considered to be main group. The group 3 elements are occasionally considered main group elements due to their similarities to the s-block elements. The group 12 elements zinc, cadmium, and mercury are sometimes regarded as main group, rather than transition group, because they are chemically and physically more similar to the p-block elements than the other d-block elements. The s-block and p-block together are usually considered main-group elements, the d-block corresponds to the transition metals, and the f-block corresponds to the inner transition metals and encompasses nearly all of the lanthanides (like lanthanum) and the actinides (like actinium). There is an approximate correspondence between this nomenclature of blocks, based on electronic configuration, and sets of elements based on chemical properties. Electronegativity is rather systematically distributed across and between blocks. Useful statements about the elements can be made on the basis of the block they belong to and their position in it, for example highest oxidation state, density, melting point . 
Austin State University) with contributing authors.The division into blocks is justified by their distinctive nature: s is characterized, except in H and He, by highly electropositive metals p by a range of very distinctive metals and non-metals, many of them essential to life d by metals with multiple oxidation states f by metals so similar that their separation is problematic. Paul Flowers (University of North Carolina - Pembroke), Klaus Theopold (University of Delaware) and Richard Langley (Stephen F. periodic table table of the elements that places elements with similar chemical properties close together pnictogen element in group 15 representative element (also, main-group element) element in columns 1, 2, and 12–18 transition metal element in columns 3–11 series (also, period) horizontal row of the period table 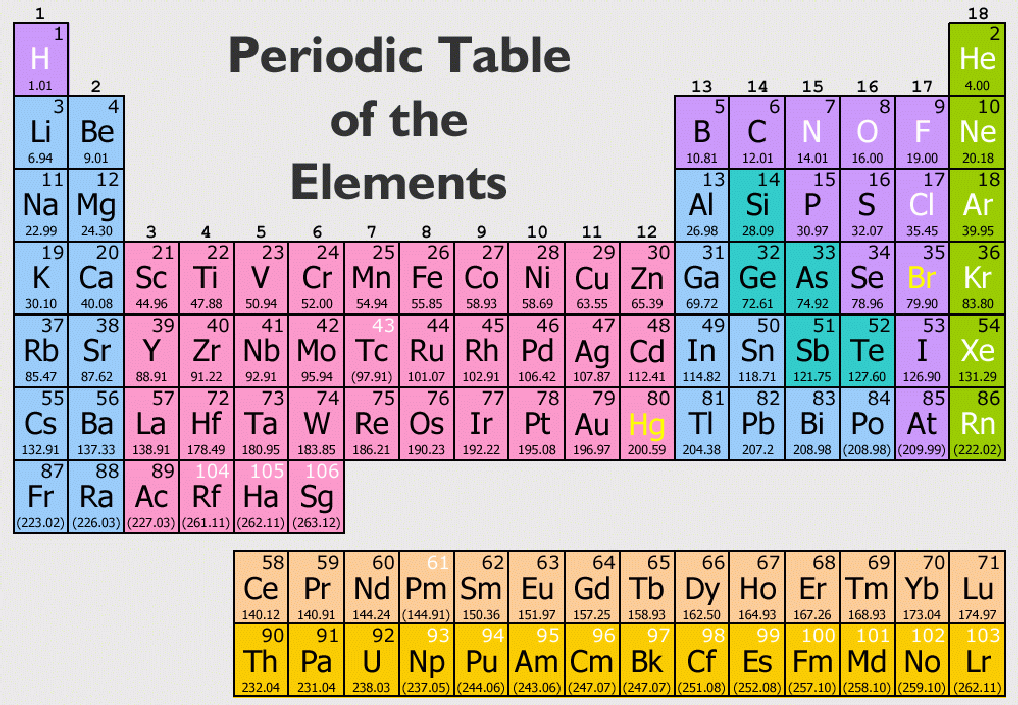
Glossary actinide inner transition metal in the bottom of the bottom two rows of the periodic table alkali metal element in group 1 alkaline earth metal element in group 2 chalcogen element in group 16 group vertical column of the periodic table halogen element in group 17 inert gas (also, noble gas) element in group 18 inner transition metal (also, lanthanide or actinide) element in the bottom two rows if in the first row, also called lanthanide, or if in the second row, also called actinide lanthanide inner transition metal in the top of the bottom two rows of the periodic table main-group element (also, representative element) element in columns 1, 2, and 12–18 metal element that is shiny, malleable, good conductor of heat and electricity metalloid element that conducts heat and electricity moderately well, and possesses some properties of metals and some properties of nonmetals noble gas (also, inert gas) element in group 18 nonmetal element that appears dull, poor conductor of heat and electricity period (also, series) horizontal row of the periodic table periodic law properties of the elements are periodic function of their atomic numbers. \): The periodic table organizes elements with similar properties into groups. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
